India to gift 500,000 doses of COVID-19 vaccine to Sri Lanka before February 4
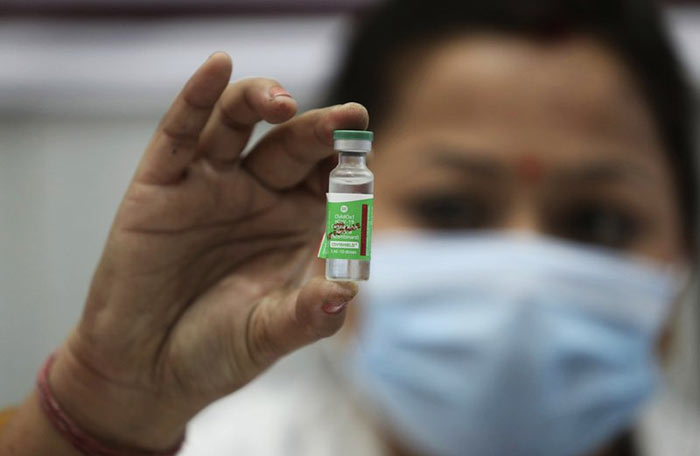
A gift of 500,000 doses of the Oxford University-AstraZeneca vaccine produced by India’s Serum Institute under the name ‘Covishield’ is expected to be airlifted to Sri Lanka to be rolled out symbolically either as early as next week to mark India’s Republic Day (January 26) or later on Sri Lanka’s Independence Day (February 4).
The National Medicines Regulatory Authority (NMRA) granted permission on Friday following an application sent by the Pune-based Serum Institute through the Indian High Commission on Tuesday.
NMRA Chairperson Prof. Asita de Silva said permission was granted on the basis of the emergency-use listing given by the drug regulatory authority of the UK (the Medicines and Healthcare products Regulatory Agency – MHRA), the pre-clinical and clinical trial data published in peer-reviewed journals and the evaluation of the dossier by two NMRA teams.
Known in the United Kingdom (UK) as the ChAdOx1 nCoV-19 vaccine (AZD1222), this vaccine has been developed by the Oxford University and is manufactured by the British-Swedish pharmaceutical company AstraZeneca. The same vaccine under the name of Covishield is being manufactured by the Serum Institute of India. This vaccine is under evaluation by the World Health Organisation (WHO) for inclusion in its emergency-use listing. So far, only the Pfizer vaccine is on this list.
“The NMRA teams looked at the clinical efficacy and safety data and product manufacture including Good Manufacturing Practice (GMP), stability and whether the parameters of the ‘lots’ produced in India match those of the products in the UK. We also took into account the fact that the UK has been administering the vaccine to its people,” said Prof. de Silva.
The vaccine is being manufactured on a large-scale at eight ‘nodes’ (sites) across the world including the Serum Institute which is registered with the NMRA.
The Serum Institute is expected to manufacture two billion doses of the vaccine this year. It produces 40 percent of the global requirements of other vaccines, it is learnt.
In preparation for the arrival of the vaccine, the Health Ministry carried out a dry run yesterday at the Piliyandala Medical Officer of Health (MOH) office, the Piliyandala Divisional Hospital and the Colombo North (Ragama) Teaching Hospital.
“We planned the minutest detail and looked into every aspect during the dry-run to fine-tune the immunisation programme,” said the Deputy Director-General of Public Health Services II, Dr. Susie Perera, adding that there might be another dry-run shortly.
Frontline health workers are at the top of the priority list for vaccinations.
The two-dose Covishield vaccine which has to be given 4-12 weeks apart as a muscular injection, has to be stored at a temperature of 2 to 8°C.
India has already gifted stocks of Covishield to Bhutan, the Maldives, Bangladesh, Nepal, Myanmar and Seychelles, while more stocks are to be sent under commercial agreements signed between Serum Institute and these countries.
Meanwhile, the Vaccine and Infectious Diseases Forum of Sri Lanka has urged that it is essential that the NMRA follow “due process” in very carefully examining the data submitted.
“Different manufacturers have different claims about their vaccines. The most credible evidence about the safety and efficacy would be what they publish specially about their Phase 3 data in peer-reviewed journals,” says the Forum.
Pointing out that only a few vaccine manufacturers have provided their data to the WHO for evaluation for prequalification for human use, the Forum states that it is only Pfizer and AstraZeneca that have published the results of Phase 3 studies and forwarded them to the WHO.
(Source: The Sunday Times – By Kumudini Hettiarachchi and Ruqyyaha Deane)

Latest Headlines in Sri Lanka
- Deputy Defence Minister reviews Coast Guard operations in Mirissa and Balapitiya May 3, 2026
- Russian Health Minister due in Sri Lanka today for official visit May 3, 2026
- Sri Lanka revises fuel prices from May 03, 2026 May 2, 2026
- Sri Lanka vehicle imports jump 900% to Rs. 195.95 Billion in Q1 2026 May 2, 2026
- Sri Lanka tourist arrivals pass 850,000 in 2026 May 2, 2026

Is this the sweetener for the Colombo Harbour East Container Terminal ?
Better pay for the goods!